Mycorrhizal Fungi: The Colonizers, Mediators, and Protectors of the Ecosystem
Posted by Daniel Schuurman on
Mycorrhizal Fungi: The Colonizers, Mediators, and Protectors of the Ecosystem
Gladis Zinati1,*, Wade Heller2, Joe Carrara3, and Amiya Kalra4
1Director of the Vegetable Systems Trial, Rodale Institute, 4 Research Technician, 611 Siegfriedale Road, Kutztown, PA 19530; 2Lead Scientist, 3Post Doc, Eastern Regional Research Center, USDA Agricultural Research Service, 600 East Mermaid Lane, Wyndmoor, PA 19038. Corresponding author’s Email Address: gladis.zinati@rodaleinstitute.org
What are mycorrhizal fungi and their benefits?
A mycorrhiza (from the Greek mykes for “fungus” and rhiza, for “root”) is a symbiotic association in which the fungus forms with plant roots. It is called symbiotic because the association benefits both organisms involved; the mycorrhizal fungi scavenge and transport otherwise unavailable nutrients to their host plants in return for carbohydrates produced through photosynthesis. These nutrients include but are not limited to phosphates, nitrates, zinc, copper, as well as organically bound nutrients (attached to carbon in large molecules that plants can’t use) that are important for plant growth. Secondary benefits of the mycorrhizal relationship with the host plant also include increased resistance to diseases, drought, and salinity.
Almost all plants growing in a wide range of ecosystems, from deserts to forests to arable land, form a symbiotic association with mycorrhizal fungi. A few plant families such as Brassicaceae (the mustard family) and Amaranthaeceae (the goosefoot family) do not form mycorrhizal associations. This fungal association should not be confused with symbiotic relationships with soil bacteria called rhizobia which result in nitrogen-fixing nodules in leguminous crops.
Types of Mycorrhizal Fungi
Two major types of mycorrhizal fungi have been described based on their structure and function: ectomycorrhizal fungi and endomycorrhizal fungi. There are two other minor fungal types that associate themselves with orchids and ericoid plants (such as blueberry, cranberry, rhododendron, and azalea).
Ectomycorrhizas (ECM) are associated with temperate-zone woody trees (such as pine, poplar, and willow). They live on the surface of the epidermal cells of the plant roots forming dense hyphae covering and branch out to the rhizosphere, but never enter the cell walls.
Endomycorrhizas, on the other hand, are associated with 80% of all plants on the planet and include arbuscular,
Endomycorrhizas are the only type of mycorrhiza that associate with herbaceous
roots (including vegetables), living inside the host’s plant root cells, forming densely
branched structures called arbuscules (See Photo 1), and thus are known as arbuscular mycorrhizal (AM) fungi.
Some species of AM fungi also form lipid storage vesicles, and thus the name vesicular arbuscular mycorrhizae (VAM) is sometimes used. The fungi grow filamentous structures out from the root into the soil environment called hyphae. The belowground mycorrhizal hyphal network expands the volume of soil that plant root system can explore and has the potential to connect plants, allowing movement of resources among plants. The richness and composition of AM fungi communities depend on the host plant, climate, and soil conditions.

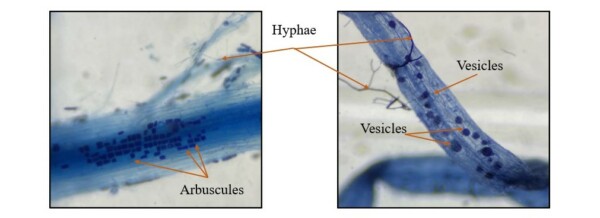
Photo 1. Structures of AM fungi on colonized of black bean roots under using trypan blue staining under microscope with 40x magnification: arbuscules, vesicles, and hyphae.
ZINATI ET AL. PUBLISHED ON SEPT 14, 2023 1
Effects of Mycorrhizal fungi on Carbon (C) Cycling
Research has shown that plants allocate between 10 and 20% of their photosynthates to AM fungi, while up to 20% and sometimes up to 50% of assimilates (organic substances made by the plant) can be allocated to ECM fungi and ericoid mycorrhizal fungi [1,2]. Almost all terrestrial and agricultural ecosystems are dominated by AM, ECM and ericoid and form symbiotic associations with trees, shrubs, vegetables, and herbs, which demonstrates that mycorrhizal fungi play a key role in global carbon cycling.
Effects of Mycorrhizal Fungi on Nitrogen (N) and Phosphorus (P) Cycles
The AM fungi contribute up to 90% of plant P to host plants in ecosystems with reduced soil P availability; their contributions of plant nitrogen (N) are less pronounced and often depend on soil type, water content, and pH [3,4]. On the other hand, ECM fungi can acquire and immobilize significant amounts of organically bound N and P in the hyphae, representing up to 80% of plant N and P.
It is important to note that not all plants investing in large amounts of C into the mycorrhizal network receive large amounts of nutrients in return. Isotope studies [5] have shown that flax plants received up to 90% of plant N and P through mycorrhizal network with little investment of C. In contrast, sorghum that invests large amounts of C receives little in terms of enhanced nutrient uptake. Such studies show an imbalance in resource exchange in mycorrhizal networks. Thus, certain plant species may benefit more from mycorrhizal networks than others.
In addition to contributing to plant nutrient uptake, mycorrhizal fungi reduce soil nutrient losses in the form of leaching or denitrification. Studies have shown that mycorrhizal fungi can significantly reduce N (up to 70 kg N/ha/yr) and P (up to 150 g P/ha/yr) leaching losses, including organic and inorganic mineral nutrients [6-9]. Thus, by minimizing nutrient losses, mycorrhizal fungi can enhance nutrient-use-efficiency and ecosystem sustainability. These services are important especially in nutrient-limited ecosystems. It has also been documented that AM fungi reduce greenhouse gas nitrous oxide (N2O) emissions by influencing bacterial communities in the rhizosphere and inducing shifts in denitrifying microbial communities [10].
Most plant roots are colonized (associated) by multiple species of mycorrhizal fungi at the same time. Several studies suggest that nutrients (e.g. N) move from one plant to another through hyphal networks [11]. This can be important for intercropping systems where N could potentially move from an N-fixing plant to a non-fixing plant.
Ecosystem Functions
Mycorrhizal fungi provide a wide range of ecosystem functions including enhancement in plant growth and productivity, seedling establishment, litter decomposition, soil formation and aggregation, and resistance to biotic and abiotic stressors (e.g. drought, heavy metals, pathogens, and pests).
Mycorrhizal fungi are known to enhance plant growth and productivity in natural and agricultural ecosystems with low but sufficient levels of P in the soil or rooting medium. Under P limitation, plants actively signal to AM fungi via root exudates to increase mycorrhizal colonization, to increase plant P concentration [12]. However, such benefit is reduced in high-input agricultural systems, particularly excess fertilizer P application. Growth responses depend on plant species; those with thick roots (such as shrubs and trees) rely more on mycorrhizal fungi than do plants with fine roots (such as grasses). Usually, plant seedlings benefit more from symbiotic association with mycorrhizal fungi than mature plants.
Almost all ecosystems are dominated by mycorrhizal associated plants except intensively managed arable fields and soils with extremely limited levels of P. Plant seedlings inoculated with AM fungi prior to transplanting into fields with low-P soils will associate effectively with AM fungi where the latter provides P and water, which are critical to plant growth and sustainability in dry conditions. Thus, inoculation with AM fungi will reduce fertilizer input, increase nutrient uptake efficiency, plant growth and yield.
As a result of fungal hyphae exploration into soil-root surroundings, the AMF brings back water and nutrients to the host plant and improves the plant’s tolerance to drought. Additional benefits include increased tolerance of host plants to soil salinity by keeping certain ions (such as Na+ and Cl-) not reaching the root system but allowing K+, Mg+2, and Ca+2. The AM fungi contribute to soil aggregation by producing a sticky sugary protein compound known as glomalin which serves as a building agent by gluing the soil particles together [13], especially the aggregate fractions >2.00mm and macroaggregates [14]. Building soil structure increases the capacity of water and nutrient retention [15]. The AM fungi increase the host resistance to soilborne diseases and pests by competition for nutrients in the rhizosphere and producing polysaccharides and phenolic compounds, thickening the plant’s cell wall, and creating a mechanical barrier to the entry of root pathogens [16].
Mycorrhizal Interaction Network
Studies have shown that AM fungi interaction networks are nested, meaning that there are several generalist fungi that associate with almost all plants. These fungi include Rhizophagus irregularis (formerly Glomus intraradices), and Funneliformis mosseae (formerly Glomus mosseae).
Root colonization of seedlings can be slow in the absence of mycelial networks, as in sites with annual plants that depends on intensively managed and tilled agricultural fields, long fallow arid environments, and sites disturbed by fire. The mycorrhizal network is low in such communities because of regular disturbance of the mycorrhizal hyphae and the vegetation cover that maintains the networks.
Factors That Lead to Reduction in Root Colonization by AM Fungi
There are many factors that lead to reduction in AM fungi plant root colonization. Below we list the common factors that contribute to soil AM fungi decline:
-
Increase in soil disturbance and intensity in both agricultural and unmanaged systems,
-
Heavy fertilization,
-
Cultivation of nonmycorrhizal crops (e.g. rapeseed, tillage radish, sugar beet) in a crop rotation where mycorrhizal association is required for next season’s crops,
-
Soils receiving runoff loaded with fungicides and herbicides, and
-
Deforestration or clearcut harvesting of forest trees which associate with AM fungi (such as maple,
ash, birch, and dogwood).
Methods to Enhance Mycelial Networks and Root Colonization
-
Reduce soil disturbance by replacing intensive tillage with reduced or no-tillage practices to provide the host plants with greater chances to associate with soil mycorrhizal fungi and to allow for a buildup of hyphae to transport nutrients and water to plants throughout the growing season.
-
Reduce the addition of fertilizer with P, especially in soils with low but sufficient levels of P. This will enhance the mycorrhizal mycelial network and increase root colonization of seedlings.
-
Inoculate seeds and plant seedlings with mycorrhizal fungi species that are compatible with host plant species. Benefits to inoculation are greatest under marginal or sub-optimal field conditions, such as highly weathered or eroded soil, low rainfall, or limited irrigation. Inoculation of seedlings prior to transplanting in the field allows the roots to form mycorrhizal associations during the seedling growth phase (often in the greenhouse), providing advantages for nutrient and water uptake which are critical at transplanting.
• Consider a crop rotation with plants that enhance indigenous mycorrhizal fungi already in the soil, such as onion, sunflower, sweet potato, potato, okra, and strawberry. Including non-mycorrhizal plants such as cabbage, broccoli, and spinach in a crop rotation may lead to low populations of indigenous mycorrhizal fungi that are needed for supporting mycorrhizal crops.
References
-
Jakobsen I., Rosendahl, L. 1990. Carbon flow into soil and external hyphae from roots of mycorrhizal cucumber plants. New Phytologist 115: 77–83.
-
Hobbie, J.E., Hobbie, E.A. 2006. 15Nin symbiotic fungi and plants estimates nitrogen and carbon flux rates in Arctic tundra. Ecology 87: 816–822.
-
Leake, J.R., Johnson, D., Donnelly, D.P., Muckle, G.E., Boddy, L., Read DJ. 2004. Networks of power and influence: the role of mycorrhizal mycelium in controlling plant communities and agroecosystem functioning. Canadian Journal of Botany 82: 1016–1045.
-
Smith, S.E., Smith, F.A. 2011. Roles of arbuscular mycorrhizas in plant nutrition and growth: new paradigms from cellular to ecosystem scales. Annual Review of Plant Biology 62: 227–250.
-
Walder, F., Niemann, H., Natarajan, M., Lehmann, M.F., Boller, T., Wiemken, A. 2012. Mycorrhizal networks: common goods of plants shared under unequal terms of trade. Plant Physiology 159: 789–797.
-
Asghari, H.R., Chittleborough, D.J., Smith, F.A., Smith, S.E. 2005. Influence of arbuscular mycorrhizal (AM) symbiosis on phosphorus leaching through soil cores. Plant and Soil 275: 181–193.
7. DOI: https://doi.org/10.21273/HORTSCI.46.5.799
-
Asghari, H.R., Cavagnaro, T.R. 2012. Arbuscular mycorrhizas reduce nitrogen loss via leaching. PLoS ONE 7:
e29825.
-
Bender S.F., Conen, F., van der Heijden, M.G.A. 2015. Mycorrhizal effects on nutrient cycling, nutrient leaching
and N2O production in experimental grassland. Soil Biol. and Biochem. 80: 283–292.
-
Bender, S.F., Plantenga, F., Neftel, A., Jocher, M., Oberholzer, H-R., Koehl, L., Giles, M., Daniell, T.J., van der Heijden, M.G.A. 2014. Symbiotic relationships between soil fungi and plants reduce N2O emissions from soil.
ISME Journal 8: 1336–1345.
-
Luo X., Liu, Y., Li, S., He, X. 2023. Interplant carbon and nitrogen transfers mediated by common arbuscular
-
mycorrhizal networks: beneficial pathways for system functionality. Front. Plant Sci. 14:1169310. Doi:
-
10.3389/fpls.2023.1169310.
14. Santos,A.,Silva,C.F.,Gama-Rodrigues,E.F.,Gama-Rodrigues,A.C.,Sales,M.,Faustino,L.L.,Barreto-Garcia, P.A.B. 2020. Glomalin in soil aggregates under different forest and pasture systems in the North of Rio de Janeiro state, Brazil. Environ. Sustain. Indic. 8, 100088.
Share this post
0 comment
